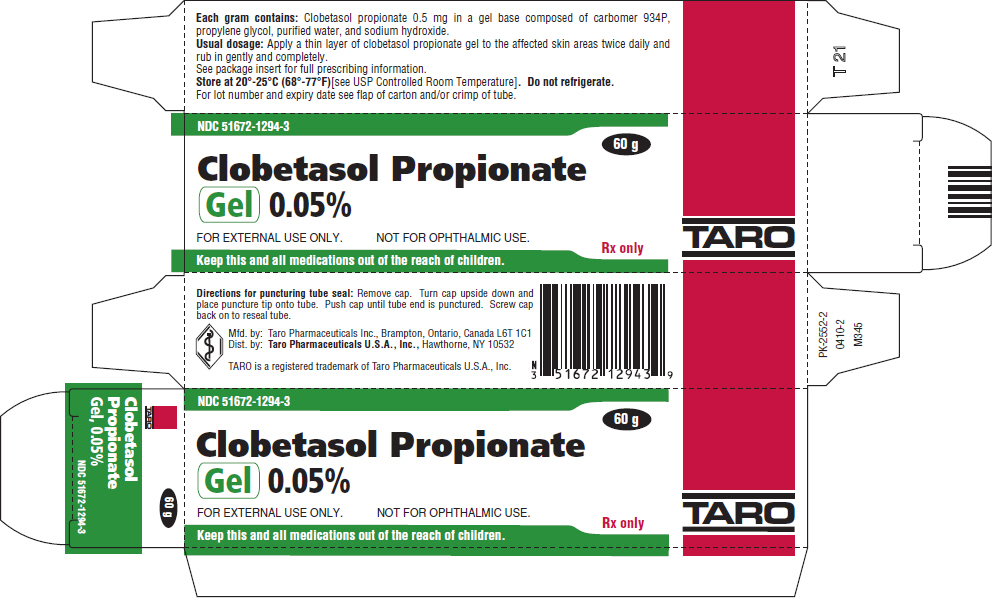
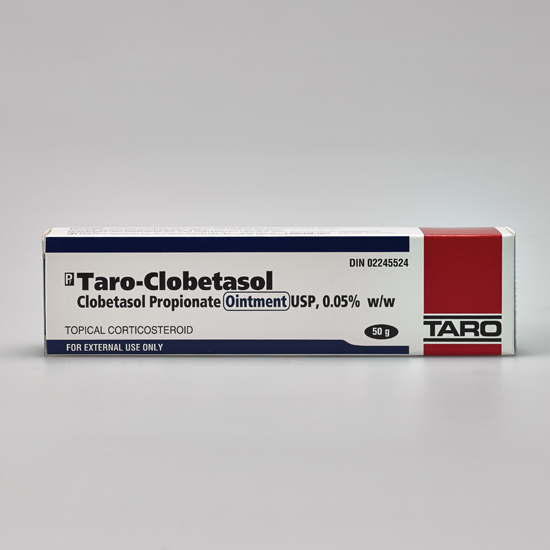
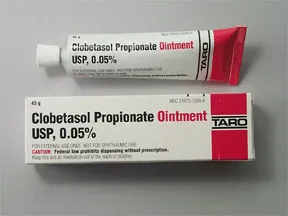
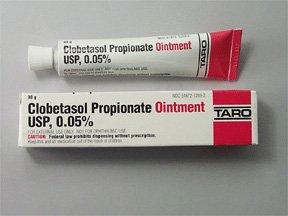
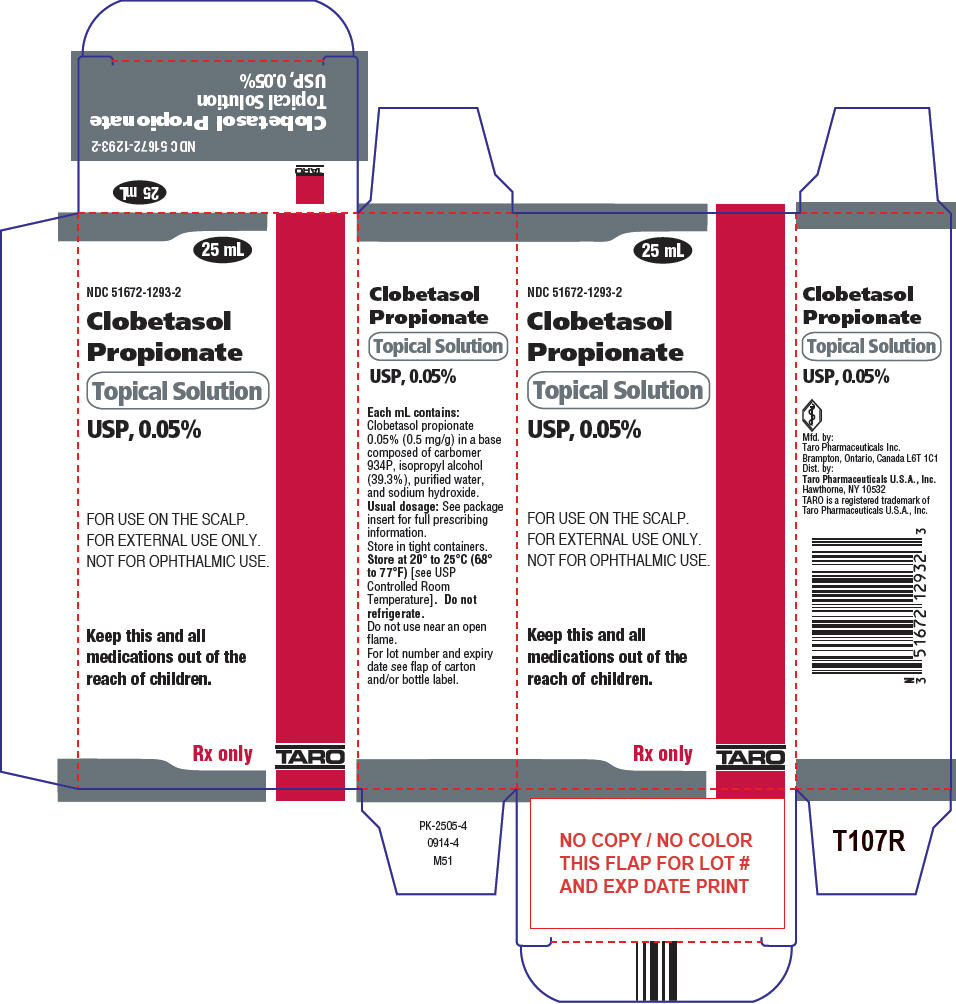
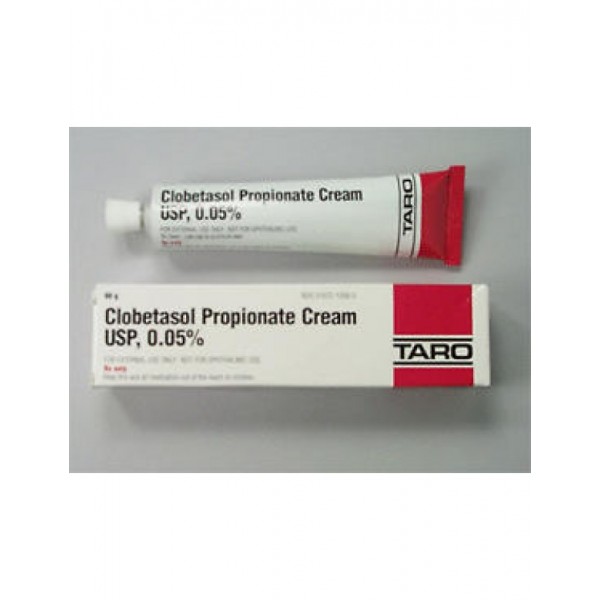
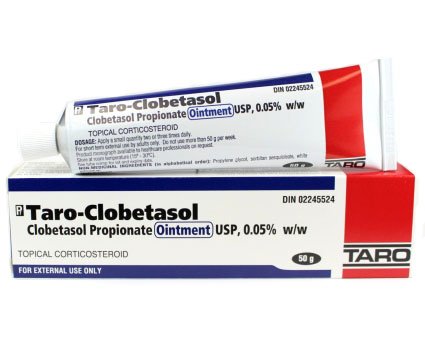
Taro Pharmaceuticals U.S.A. Issues Voluntary Nationwide Recall of Clobetasol Propionate Ointment USP, 0.05%, 60 g Tubes, Lot AC13786 Due to Microbial Contamination | FDA
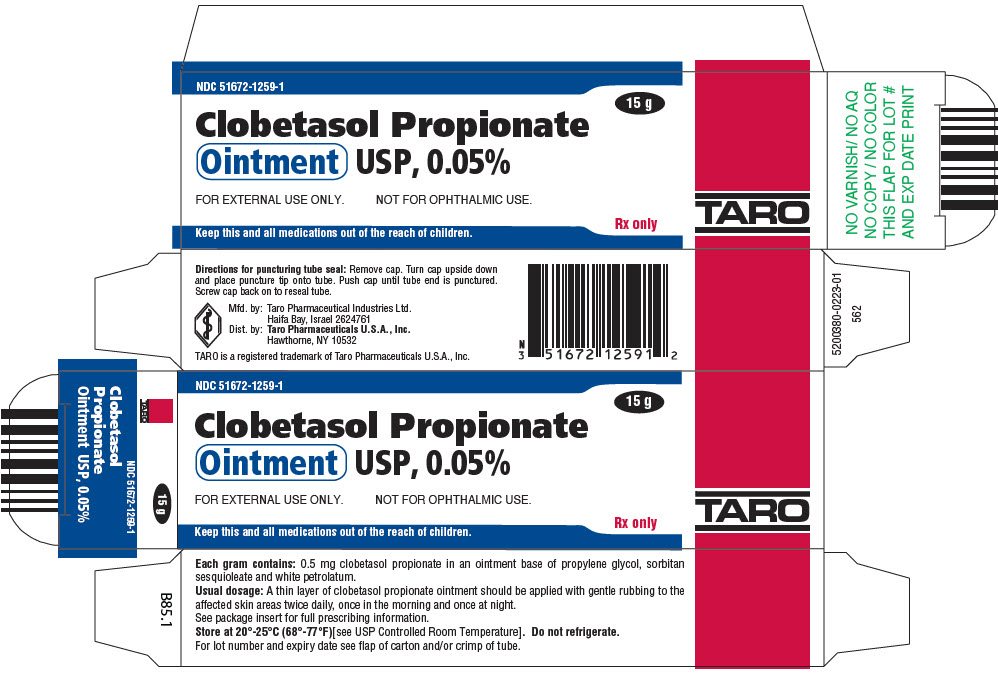
Clobetasol Propionate Gel, 0.05% Clobetasol Propionate Cream USP, 0.05% Clobetasol Propionate Ointment USP, 0.05%